Quantitatively assessing behaviour to measure animal health is challenging because there is a lack of unobtrusive behavioural models to refine animal testing procedures. LocoWhisk is a novel arena and software solution that simultaneously measures locomotion behaviours and whisker movements, providing robust and quantitative measures of rodent behaviour for basic research or drug discovery.
The LocoWhisk system is a new, portable behavioural set-up that incorporates both gait analysis (using a pedobarograph) and whisker movements (using high-speed video camera and infrared light source). The system has so far been successfully piloted on many rodent models, and would benefit from further validation and commercialisation opportunities.
Measurable behavioural models have been proposed as a way to mitigate stress in animal testing, allowing the animal to be studied while behaving freely. Several behavioural models have been developed, such as the Morris Water Maze, Rotorod and grip strength task, but these require extensive animal training (which can be very stressful for the animals), and usually only measure the duration and frequency of certain behaviours. Most laboratories will expose their animals to a host of these behavioural tests in order to capture a range of behaviours, requiring training for each individual test and repeated testing of each individual animal. Locomotion testing can be more measureable, but still requires extensive animal training (i.e. Noldus catwalk).
LocoWhisk overcomes these challenges by simultaneously measuring whisker movements and locomotion to give an added dimension to quantifying motor control (Grant et al., 2012). Many small laboratory mammals (rats, mice, guinea pigs, shrews, opossums) rely on their whiskers as their primary sense to guide locomotion and environmental exploration (Grant et al., 2009; 2014; 2016a; 2016c). LocoWhisk developers have characterised rodent whisker movements (Grant et al., 2009; Mitchinson et al., 2007) and found that aspects of whisker movements can be reliably interrupted during a variety of cognitive and motor disorders, including rodent models of motor neuron disease (Grant et al., 2014), anxiety (Grant et al., 2016b), Huntington’s disease (Garland et al., 2017), Parkinson’s disease (Grant et al., in prep) and dementia (Grant et al., in prep). This demonstrates the utility of measuring aspects of whisker movements to quantify motor and cognitive decline. It has also been demonstrated that whisker movements can be linked to an animal’s locomotion speed, and also adjusted according to their environmental experience and attention (Arkley et al., 2014).
References:
- Arkley K, Grant RA, Mitchinson B, et al. (2014). Strategy change in vibrissal active sensing during rat locomotion. Current Biology 24(13): 1507-1512. doi: 10.1016/j.cub.2014.05.036.
- Garland H, Wood NI, Skillings EA, et al. (2017). Characterisation of progressive motor deficits in whisker movements in R6/2, Q175 and Hdh knock-in mouse models of Huntington’s disease. J. Neuroscience Methods 300: 103-111. doi: 10.1016/j.jneumeth.2017.04.020.
- Grant R A, & Arkley K P (2016a). Matched Filtering in Active Whisker Touch. In: The Ecology of Animal Senses (Ed. von der Emde G, Warrant E), Springer International Publishing. doi: 10.1007/978-3-319-25492-0_3.
- Grant RA, Cielen N, Maes K, et al. (2016b). The effects of smoking on whisker movements: A quantitative measure of exploratory behaviour in rodents. Behavioural processes 128: 17-23. doi: 10.1016/j.beproc.2016.03.021.
- Grant RA, Delaunay MG, & Haidarliu S (2016c). Mystacial whisker layout and musculature in the guinea pig (Cavia porcellus): A social, diurnal mammal. The Anatomical Record 300(3): 527-536. doi: 10.1002/ar.23504.
- Grant RA, Haidarliu S, Kennerley NJ, et al. (2013). The evolution of active vibrissal sensing in mammals: evidence from vibrissal musculature and function in the marsupial opossum Monodelphis domestica. Journal of Experimental Biology 216(18): 3483-3494. doi: 10.1242/jeb.087452.
- Grant RA, Mitchinson B, Fox CW, et al. (2009). Active touch sensing in the rat: anticipatory and regulatory control of whisker movements during surface exploration. Journal of neurophysiology 101(2): 862-874. doi: 10.1152/jn.90783.2008.
- Grant RA, Mitchinson B, & Prescott TJ (2012). The development of whisker control in rats in relation to locomotion. Developmental psychobiology 54(2): 151-168. doi: 10.1002/dev.20591.
- Grant RA, Sharp PS, Kennerley AJ, et al. (2014). Abnormalities in whisking behaviour are associated with lesions in brain stem nuclei in a mouse model of amyotrophic lateral sclerosis. Behavioural brain research 259: 274-283. doi: 10.1016/j.bbr.2013.11.002.
- Mitchinson B, Martin CJ, Grant RA, et al. (2007). Feedback control in active sensing: rat exploratory whisking is modulated by environmental contact. Proceedings of the Royal Society of London B: Biological Sciences 274(1613): 1035-1041. doi: 10.1098/rspb.2006.0347.
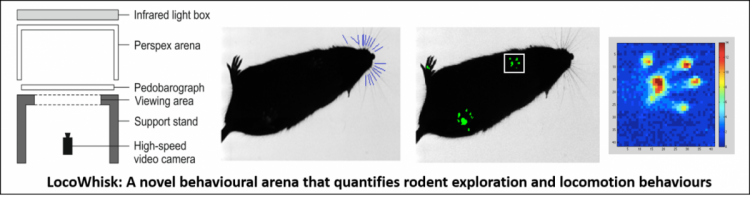
The LocoWhisk arena records quantitative behaviours (whisker movements and locomotion), using a brand new experimental set-up. It utilises a high-speed video camera, pedobarograph and infrared lighting technologies. The arena is compatible with standard high-speed cameras that are present in many behavioural testing laboratories; enabling behaviours to be reliably measured using state-of-the-art image processing methods (Hewitt et al. 2017; Sherwood et al. 2016). An in-house, open source and freely available locomotion and whisker tracker has been developed to wholly automatically extract whisker movements from the collected video footage using image processing techniques. Movement measures include estimates of angle, frequency, spread, amplitude, speed, curvature and asymmetry, which are highly quantitative and have been found to change under a range of health conditions.
It has so far been successfully piloted on adult mice (see Figure 1) and rats; however, it now needs to be further used, adapted and validated on a number of mouse models and experimental set-ups, to ensure maximal user uptake and satisfaction. The primary ambition of the LocoWhisk arena is to provide a new, non-invasive assessment of animal health and welfare. The entirely novel aspect of this system is the use of whisker measurements, which have been found, in the case of Huntington’s disease mice (Garland et al. 2017), to reveal motor deficits earlier than any other behavioural test, including gait assessments. This is particularly useful for quantifying new animal models of disease, and the therapeutic benefit of new drugs.
While the LocoWhisk arena and associated software has its roots strongly embedded in measuring animal behaviour non-invasively, it could also incorporate electrophysiology and electromyography equipment. This would be interesting to explore further with an interested partner. This means that musculoskeletal or neural measurements can be collected alongside behavioural assessments, to quantify physiological aspects of animal health. In addition, the system is in no way limited to rodents. The locomotion aspect of the system, especially, can be amended to include a range of animals. Therefore, although the system was developed specifically to focus on the assessment of health and welfare of small laboratory mammals, future studies could include locomotion assessments of larger laboratory, farm and domestic animals.
Figure 1. The LocoWhisk Arena. Consists of an integrated high-speed filming arena and pedobarograph (far left), so that both whiskers and feet can be clearly imaged and tracked (centre), to give quantitative measures of motor control, such as force distribution plots of the feet for gait analysis (right).
References:
- Garland H, Wood NI, Skillings EA, et al. (2017). Characterisation of progressive motor deficits in whisker movements in R6/2, Q175 and Hdh knock-in mouse models of Huntington’s disease. J. Neuroscience Methods 300: 103-111. doi: 10.1016/j.jneumeth.2017.04.020.
- Hewitt B, Yap MH, Hodson-Tole EF, et al. (2017). A novel automated rodent tracker (ART), demonstrated in a mouse model of amyotrophic lateral sclerosis. J. Neuroscience Methods 300: 147-156. doi: 10.1016/j.jneumeth.2017.04.006.
- Sherwood T, Ahmad E, & Yap MH (2016). Formulating Efficient Software Solution for Digital Image Processing System. Journal of Software: Practice and Experience 46(7): 931-954. doi: 10.1002/spe.2339.
Partnerships are being sought with academic or industry collaborators that have interesting small laboratory mammal models to test and validate the LocoWhisk system further. Models with motor control deficits, such as stroke, Parkinson’s disease and aging are of particular interest. In addition, quantifying the effect of a new treatment strategy or drug on an established animal model would help to further validate the LocoWhisk system.
Partnerships with collaborators that would like to make use of the LocoWhisk arena in their lab in tandem with their own specific system, such as with EMG or electrophysiology recordings would also be of interest. The LocoWhisk system could be lent to these labs to trial, or for longer, more specific studies a new arena would be developed at a reduced cost. Partners would work together to develop and validate the system and then publish the findings.
Partnerships with consumer product, biotechnology or laboratory facilities companies interested in commercialising the LocoWhisk arena and software, or combining it with existing recording devices, such as electrophysiology, would also be of interest.
Uptake of the LocoWhisk tool by academic researchers and industry will reduce animal use in small mammal behavioural testing, by enabling the same animal to be observed over the duration of the experiment, or disease progression.
So far, this approach has significantly improved upon traditional behavioural methods by:
- Making sure all animals complete the task (none lost due to not being able to learn).
- Reducing stress in animals by having short experimental times (10 mins) and enabling experiments to be conducted in the dark.
- Generating wholly quantitative data using specialist image-processing software that can measure very precise aspects of motor control from short movement bouts.
- Reducing severity of disease symptoms by diagnosing motor deficits earlier in disease progression.
- Reducing the number of animals in each condition, as significant volumes of quantitative data can be collected from each animal. Indeed, significant results can be seen between groups containing only five animals (Grant et al. 2014), rather than the large numbers that are usually recommended for behavioural testing (12 animals of single sex, Ludolph et al., 2010).
- The ability to simultaneously measure whisker movements and locomotion offers substantial 3Rs benefits through both the increased sensitivity of the dual testing and the reduced time for experimental procedures.
Alongside the LocoWhisk arena, a new LocoWhisk data sharing platform is being developed to enable researchers to reanalyse data generated in the LocoWhisk.
References:
- Grant RA, Sharp PS, Kennerley AJ, et al. (2014). Abnormalities in whisking behaviour are associated with lesions in brain stem nuclei in a mouse model of amyotrophic lateral sclerosis. Behavioural brain research 259: 274-283. doi: 10.1016/j.bbr.2013.11.002.
- Ludolph AC, Bendotti C, Blaugrund E, et al. (2010). Guidelines for preclinical animal research in ALS/MND: a consensus meeting. Amyotroph Lateral Scler 11(1–2): 38–45. doi: 10.3109/17482960903545334.