UnTangle
The aim of this Challenge was to develop a physiologically relevant human stem cell-derived neuronal assay to predict the efficacy and unexpected pharmacological effects of new chemical entities and biologics targeting tau in Alzheimer’s disease.
To solve this Challenge, the awarded team led by Dr Selina Wray at University College London, have adapted iPSC-neurons and cultured them in microfluidic devices for the study of neuronal connectivity and the spread of protein pathologies between synaptically connected chambers.
Publication
Karikari TK et al. (2019). Distinct Conformations, Aggregation and Cellular Internalization of Different Tau Strains. Frontiers in Cellular Neuroscience 13:296. doi:10.3389/fncel.2019.00296
This work was funded through a Phase 1 award.
Publication
Ritchie L et al. (2018). Toll-like receptor 3 activation impairs excitability and synaptic activity via TRIF signalling in immature rat and human neurons. Neuropharmacology 135, 1-10. doi:10.1016/j.neuropharm.2018.02.025.
Challenge completed
The UnTangle CRACK IT Challenge led by Dr Selina Wray has been successfully completed and the team adapted iPSC-neurons and cultured them in microfluidic devices for the study of neuronal connectivity and the spread of protein pathologies between synaptically connected chambers.
Invited speaker
Dr Selina Wray was an invited speaker at the EuroTau Meeting (Lille, France)
Conference presentation
Miniaturized Systems for Chemistry and Life Sciences (Dublin, Ireland)
Dementia on a chip: Monitoring synaptic dysfunction of patient derived human neurons.
Publication
Arber C, Lovejoy C and Wray S (2017). Stem cell models of Alzheimer’s disease: progress and challenges. Alzheimer's research & therapy 9(1): 42. doi:10.1186/s13195-017-0268-4.
Invited speaker
Alzheimer’s Research UK 2017 Annual Conference (Aberdeen, UK)
Conference presentation
Alzheimer's & Parkinson's Diseases Congress 2017 (Vienna, Austria)
Tau phosphorylation and splicing in iPSC neurons from AD and FTD.
Invited speaker
Dr Selina Wray was an invited speaker at the Scottish TSE Network Annual Symposium (Edinburgh, UK)
Neurodegeneration and the 3Rs Models, Mechanisms & Resources.
Conference presentation
Miniaturized Systems for Chemistry and Life Sciences (Dublin, Ireland)
Dementia on a chip: Monitoring synaptic dysfunction of patient derived human neurons.
Public engagement
Dr Graham Robertson presented at Images of Research, University of Strathclyde
Publication
Sposito T et al. (2015). Developmental regulation of tau splicing is disrupted in stem cell-derived neurons from frontotemporal dementia patients with the 10 + 16 splice-site mutation in MAPT. Hum Mol Genet 24(18):5260-9. doi:10.1093/hmg/ddv246.
Publication
Wray S et al. (2015). Higher Mitochondrial Membrane Potential Induces ROS Production in the Familiar Form of Frontotemporal Dementia with MAPT Mutations. Biophysical Journal. doi:10.1016/j.bpj.2014.11.3324.
Conference presentation
Miniaturized Systems for Chemistry and Life Sciences (San Antonio, USA)
A microfluidics platform for human tau mutation neurons.
Phase 2 awarded
A team led by Dr Selina Wray, University College London, has been awarded £898,416 to deliver the project: Untangling tauopathies: A microfluidics based drug-screening platform for human tau mutation neurons.
NC3Rs blog post
Working together to CRACK dementia.
Phase 1 awarded
Four Phase 1 Awards were made to project teams led by:
- Dr Eric Hill, University of Aston, £95,453.
- Professor Maria Grazia Spillantini, University of Cambridge, £66,898.
- Dr Selina Wray, University College London, £99,956.
- Dr Christopher Ward, University of Manchester, £81,956.
Challenge launched
Sponsored by Alzheimer's Research UK, Lilly and Janssen, the UnTangle Challenge aims to develop a physiologically relevant human stem cell-derived neuronal assay to predict the efficacy and unexpected pharmacological effects of new chemical entities and biologics targeting tau in Alzheimer’s disease.
Background
One of the most active areas of research into Alzheimer’s disease centres on the tau protein that forms the classic neurofibrillary tangles. Aberrant intracellular inclusions composed of hyperphosphorylated filamentous tau are not only a neuropathological hallmark of Alzheimer’s disease, but are also the key pathological species in progressive supranuclear palsy and other sporadic neurodegenerative disorders, which have been collectively termed tauopathies.
The discovery that pathogenic mutations in the tau gene microtubule associated protein tau (MAPT) can cause a familial neurodegenerative tauopathy has provided compelling evidence that tau dysfunction is sufficient to cause neurodegeneration. Many recent in vivo studies have shown that tau aggregates have ‘prion-like’ properties which not only allow them to transmit or seed further tau aggregation, but also spread to neighbouring cells or functionally connected brain regions. This process is referred to as ‘tau propagation’ and might explain the stereotypic progression of tau pathology in the brains of Alzheimer’s disease patients.
Study of the development and spread of tau pathology in animal models requires large numbers, is time consuming and expensive. Often, the mechanisms and/or potential drug targets that are identified do not translate into humans.
3Rs benefits
Thousands of transgenic mice are used per year in large pharmaceutical companies with additional breeding animals to obtain the required genotype. There are specific challenges associated with tau transgenics:
- Phenotypic variability. Mice often undergo chronic treatment with a candidate drug to test for disease modifying actions. Large numbers of animals are required per treatment arm to ensure clear results. For example, Lilly have noted wide phenotypic variability in mice expressing mutant human tau (JNPL3) leading to n=25 animals per treatment arm being required for drug studies.
- Variability in levels of tau expression. Some transgenic lines express varying levels of tau and/or only a certain portion of mice are suitable for drug testing (e.g. only 25% of tg4510 mice are bigenic).
Therefore, large breeding programmes are needed to generate sufficient numbers of mice for each study. A human cell-based assay would reduce the number of transgenic animals used to investigate tau pathology and also improve the research and development of new treatments in this area.
Phase 1 winners
Project teams led by:
- Dr Eric Hill, University of Aston, £95,453.
- Professor Maria Grazia Spillantini, University of Cambridge, £66,898.
- Dr Selina Wray, University College London, £99,956.
- Dr Christopher Ward, University of Manchester, £81,956.
Phase 2 winner
Project team led by:
- Dr Selina Wray, University College London, £898,416.
Full Challenge information
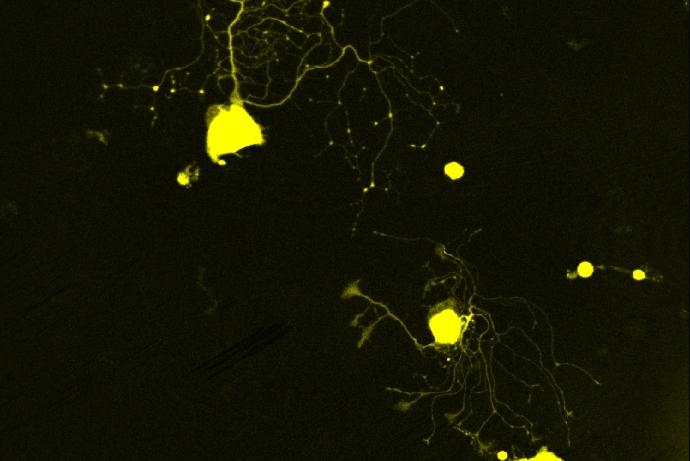
A multidisciplinary team led by Dr Selina Wray (University College London) has generated human induced pluripotent stem cell-derived neurons (hiPSC-neurons) carrying mutations in the tau gene and isogenic controls, cultured in microfluidic devices, to study neuronal connectivity and the spread of tau pathologies between synaptically connected chambers. This provides a human-relevant model of tauopathy, reducing the number of animals used to study tau pathology and for drug screening in the development of novel therapeutics.
HiPSC-neurons are seeded into environmentally isolated chambers in microfluidic devices. HiPSC-neurons cultured in the microfluidic devices form active synaptically connected networks, and the uptake and spread of tau between connected chambers can be detected using exogenous tau fibril seeds.
The team demonstrated that after long term culture (365 days), control neurons express all six tau isoforms seen in the adult human central nervous system, and that neurons with mutations in tau show dysregulated tau splicing (Sposito et al., 2015).
The team has developed methods to cryopreserve human hiPSC-dervied neuronal precursors directly within the microfluidic devices, with limited impact on cell viability or neuronal function. This offers the opportunity for product development to enable potential customers to purchase microfluidics kits with neurons ready plated within a device, which can be revived, cultured until maturity and used to study synaptic connectively and the spread of protein pathologies. The team are currently exploring options to support further development of this model prior to commercialisation.
For further information about the model please contact Dr Selina Wray
Human stem cell model of tau pathology at EBiSC
Quality assured human induced pluripotent stem cell (hiPSC) lines carrying mutations in the tau gene (MAPT) are available for use by the research community, pharmaceutical industry and other organisations via a central repository – the European Bank of induced Stem cells (EBiSC).
Neurons produced by these cell lines provide a human-relevant model to study tau pathology and for drug screening of novel therapeutics, reducing reliance on animal models.
A total of 13 lines have been deposited with cell line names SIGi001-A-1 to SIGi001-A-13.
Access the technology
Find out more about the model and how to access it on the EBiSC website.