International 3Rs Prize celebrates animal-free research

An animal-free organoid model has won the 3Rs Prize, awarded by the NC3Rs and co-funded by GSK.
This year’s 3Rs Prize was presented at an online award ceremony on Thursday 20 July. Attendees heard from the winner, Dr Lisa Wagar, about how she aims to streamline vaccine development for global respiratory pathogens of concern using her entirely animal-free tonsil organoid model. Professor Alex Frangi and Dr Ali Sarrami-Foroushani also presented the first ever in silico trial for a medical device from their highly commended paper. You can watch the recording of the webinar on our YouTube channel:
Read more about the winning research:
- Winner – animal-free organoids for respiratory infection and immunity
- Highly commended – virtual patients for in silico clinical trials
"Every year the Panel has the difficult decision selecting a winner from a wealth of innovative and impactful papers. The applications this year reflected the high standard of exciting 3Rs research taking place worldwide. The winning paper presents an elegant 3Rs solution to meet a significant and pressing need and I look forward to seeing the continued adoption of this replacement method in labs around the world."
Professor Kevin Shakesheff, Chair of the NC3Rs Board and 3Rs Prize Panel
"This is the 18th year that GSK has supported the NC3Rs 3Rs International Prize. We are delighted to see that the quality of the winning entries continues to be truly impressive and pivotal to scientific innovations. Our commitment to the 3Rs prize aligns with our strategic direction and our 3Rs ambition being integral to our purpose of developing medicines and vaccines. Where possible we utilise non-animal methods and technologies, and we continue to invest in novel experimental systems based on human cells and tissues that hold promise of further reducing the need for animal experimentation. When animal research is conducted or commissioned, we are seeking ways to minimise animal use and reduce the impact on the animals. We know how impactful this prize is to the winners and look forward to seeing how their careers develop and the impact their research will make on the wider 3Rs community."
Dr Joanne Storey, Executive Director, GSK 3Rs Strategy Lead
Winner – animal-free organoids for respiratory infection and immunity

Dr Lisa Wagar from the California Irvine University, USA, has been awarded the 3Rs Prize for her entirely animal-free tonsil ‘organoid’ model of respiratory infection and immunity. Respiratory infections are the deadliest communicable disease and the fourth leading cause of death globally, even before the COVID-19 pandemic. Research still largely relies on animal models which may poorly replicate the complex responses to infection and vaccination in humans. Lisa used cells from human tissue samples to develop miniature tonsils, called organoids, to investigate the immune response to infection and vaccination.
In the winning paper, Lisa uses the system to investigate how immunity develops in response to flu, MMR and COVID-19 vaccination. The organoids were able to capture individual patient variation in somatic hypermutation, affinity maturation and B cell class switching. This is the first time these aspects of the adaptive immune response have been accurately reflected in an in vitro system, replacing the use of animals with a more human relevant model. Lisa’s organoids do not use any animal cells or animal-derived products such as cell culture media/sera or antibodies.
The paper has been viewed over 53,000 times and Lisa’s model is already being used by international collaborators. The Prize grant will support early careers researchers in the Wagar lab to present their research at conferences, showcasing the utility of the model and driving its adoption alongside building the future skills base in non-animal organoid approaches. By developing the technology into a high throughout system that can handle more samples in less time, Lisa aims to maximise the efficiency of respiratory disease research, streamline vaccine development and tackle global pathogens of concern, all whilst replacing the use of animals.
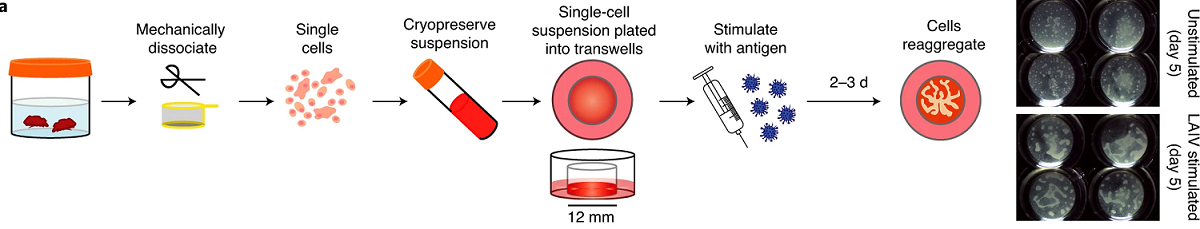
View a high resolution version of the graphical abstract.
Read the paper: Wagar LE et al. (2021). Modeling human adaptive immune responses with tonsil organoids. Nat Med 27:125-135. doi.org/10.1038/s41591-020-01145-0
Highly commended – virtual patients for in silico clinical trials

Professor Alex Frangi and Dr Ali Sarrami-Foroushani at the University of Leeds were highly commended for their report of the first in silico clinical trial for a medical device. Their virtual patient populations accurately replicated and expanded on findings from clinical trials of devices to treat brain aneurysms, reproducing results that took decades and over 4,000 animals to produce in a matter of days.
The highly commended paper exemplifies how in silico trials have the potential to replace the use of animals by accurately predicting the safety and efficacy of medical devices and drugs. Research into treating vascular diseases of the brain (e.g. aneurysm and stroke) typically uses large animals including pigs, dogs and rabbits. Between 2000 and 2015, more than 430 rabbits and 160 dogs were used in studies focusing on the flow-diverting stents that Alex and Ali modelled in silico. Virtual trials can provide evidence to ensure only treatments with the highest likelihood of clinical benefit and lowest risk of complications progress to later-stage testing, avoiding the use of animals in preclinical safety studies.
In addition, Alex and Ali’s in silico model goes beyond what is possible in conventional animal and human trials. Their virtual trials were able to assess an individual patient’s risk of complications and treatment failure, supporting clinical decisions to be made on a case-by-case basis. The model’s virtual population approach has broad potential applications across disciplines, for example in cardiovascular (heart valves and pacemakers) and musculoskeletal (hip and knee replacements) medicine. The paper adds to the body of reliable regulatory evidence that is critical to support the adoption of in silico testing and trials in the UK and internationally. It was a key driver in establishing the in silico medicine innovation network (InSilicoUK), a community of scientists, learned societies and SMEs brought together by Innovate UK to advance the use of in silico approaches which replace the use of animals and improve patient outcomes.
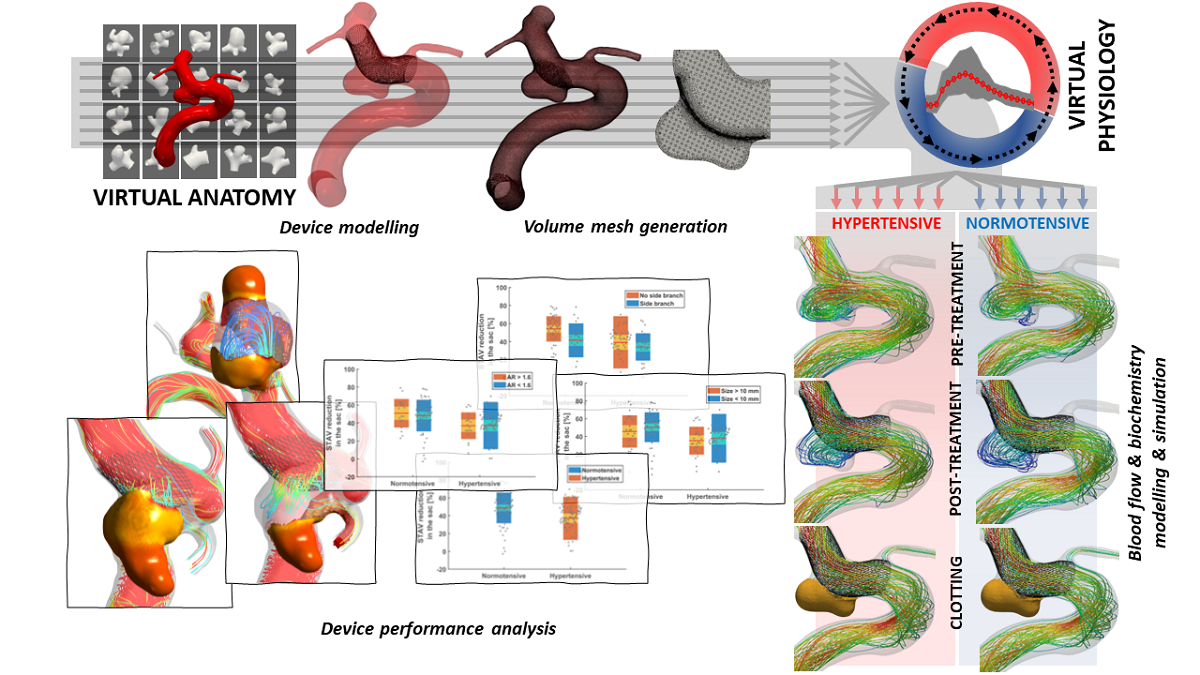
View a high resolution version of the graphical abstract.
Read the paper: Sarrami-Foroushani A et al. (2021). In-silico trial of intracranial flow diverters replicates and expands insights from conventional clinical trials. Nat Commun 12:3861. doi.org/10.1038/s41467-021-2 3998-w
About the 3Rs Prize
Co-funded by GSK, the 3Rs Prize is awarded annually by the NC3Rs for a paper that describes outstanding and original work that has or could have major impacts on the replacement, reduction or refinement of the use of animals in research. The competition is open to any research team in the world and applications are assessed by an expert Panel.
The winning researcher was awarded a £28k grant and a £2k personal award. The highly commended winner receives a £4k grant and a £1k personal award.
About GSK
GSK is a global biopharma company with a purpose to unite science, technology, and talent to get ahead of disease together. Further information can be found on GSK’s website.
About the NC3Rs
The National Centre for the Replacement, Refinement and Reduction of Animals in Research (NC3Rs) is a UK-based scientific organisation dedicated to helping the research community worldwide to replace, refine and reduce the use of animals in research. Primarily funded by the UK Government, the NC3Rs is also supported by the charitable and private sectors. It collaborates with organisations from across the life sciences sector, nationally and internationally, including universities, industries, other research funders and regulatory authorities. Further information can be found on the NC3Rs website.
