Blood sampling: Marmoset
Approaches for sampling blood in the marmoset, covering non-surgical and terminal techniques.
General principles
You should read the general principles of blood sampling page before attempting any blood sampling procedure.
Femoral vein (non-surgical)
Technique
Common marmosets can be trained to cooperate with blood sampling thus reducing associated stress. They will remember receiving a reward (e.g. food treat) after the procedure, which can make them easier to handle during subsequent procedures.
Sampling from the femoral vein is the most commonly used route in the marmoset. The vein is not usually visible and stemming blood flow can be slow. The femoral triangle is prone to bruising. Analgesia can be used if necessary.
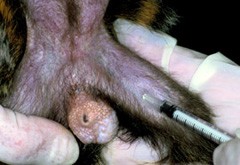
The technique should be carried out aseptically. Where multiple samples are taken alternate legs should be used. 0.1 - 3 ml of blood can be obtained per sample and, depending on sample volume and scientific justification usually no more than seven samples in a 24-hour period should be taken. The number of attempts to take a blood sample should be minimised (no more than three needle sticks in any one attempt) and the femoral triangle should be allowed to recover before the next blood sampling session. Finger pressure on cotton wool should be applied to the site until bleeding stops, after which the marmoset can be returned to its cage.
Catching and restraining a marmoset can cause it stress, especially for repeated sampling. Training the animal to enter a restraining box or cage turret can reduce the stress of capture. The duration of restraint should be kept to a minimum. Good preparation of the area where the sampling will take place will help to minimise the time taken to complete the procedure.
Summary
| Number of samples | No more than seven blood samples should be taken in any 24-hour period. |
| Sample volume | Up to 0.1-3 ml |
| Equipment | 25G needle |
| Staff resource | Two people are required: one to take the blood sample and another to restrain the marmoset. |
| Adverse effects | Haematoma if blood flow is not stopped. |
| Other | The femoral triangle is prone to bruising and stopping the blood flow can be slow. |
Resources and references
- Morton DB et al. (1993). Removal of blood from laboratory mammals and birds. Lab Animal 27(1): 1-22. doi: 10.1258/00236779378108241
- Diehl KH et al. (2001). A good practice guide to the administration of substances and removal of blood, including routes and volumes. Journal of Applied Toxicology 21(1): 15-23. doi: 10.1002/jat.727
- Schultz-Darken NJ (2003). Sample collection and restraint techniques used for common marmosets (Callithrix jacchus). Comparative Medicine 53(4): 360-3. PMID: 14524411
- Lucas RL et al. (2004). Collection and preparation of blood products. Clinical Techniques in Small Animal Practice 19(2): 55-62. doi: 10.1053/j.ctsap.2004.01.007
- European Association of Zoos and Aquaria (2017). Best Practice Guidelines for Callitrichidae. 3.1 Edition. EAZA. (p. 160). Available at: www.eaza.net/assets/Uploads/CCC/2017-Callitrichidae-EAZA-Best-Practice-Guidelines-Approved.pdf
- Hopper J (2020). Common Marmoset. In: Handbook of Exotic Pet Medicine (Ed. Kubiac M), 1st edition. John Wiley & Sons Ltd.
Abdominal vena cava (terminal)
Technique
This is a suitable method to obtain a single, large, good quality blood sample when the marmoset is under terminal anaesthesia or euthanised, as the body cavity must be opened to expose the blood vessel.
A laparotomy is performed and viscera are moved to one side to identify the abdominal vena cava. Surrounding connective tissue and fat are then removed to expose and isolate the vessel. Application of finger pressure to the blood vessel can help dilate the vessel. Depending on the size of the marmoset, up to 10 ml of blood can be collected. Care should be taken to withdraw the blood slowly to prevent the vessel collapsing.
Summary
| Number of samples | One |
| Sample volume | Up to 10 ml |
| Equipment | 19G needle |
| Staff resource | Two people are required: one to take the blood sample and another to perform the anaesthesia (unless carried out on a euthanised animal). |
You should read the general principles of blood sampling page before attempting any blood sampling procedure.