Mini-gut organoid model reveals the early stages of whipworm infection
NC3Rs-funded research could replace mice in tropical disease research.
Read the full paper: Defining the early stages of intestinal colonisation by whipworms (Duque-Correa et al., 2022).
Researchers have developed the first in vitro system to study how the gut lining responds to whipworm infection. The work, part-funded by an NC3Rs Fellowship awarded to Dr María Duque-Correa at the Wellcome Sanger Institute, is now published in Nature Communications.
Trichuriasis, the disease caused by the whipworm parasite Trichuris trichiura, is a neglected tropical disease that affects as many as 500 million people worldwide, most commonly in tropical and subtropical regions. Chronic whipworm infections can cause a range of debilitating gastrointestinal issues and nutritional deficiencies, and are linked to delays in physical and cognitive development, especially in children. There are few treatments available, and they cannot completely eradicate the infection.
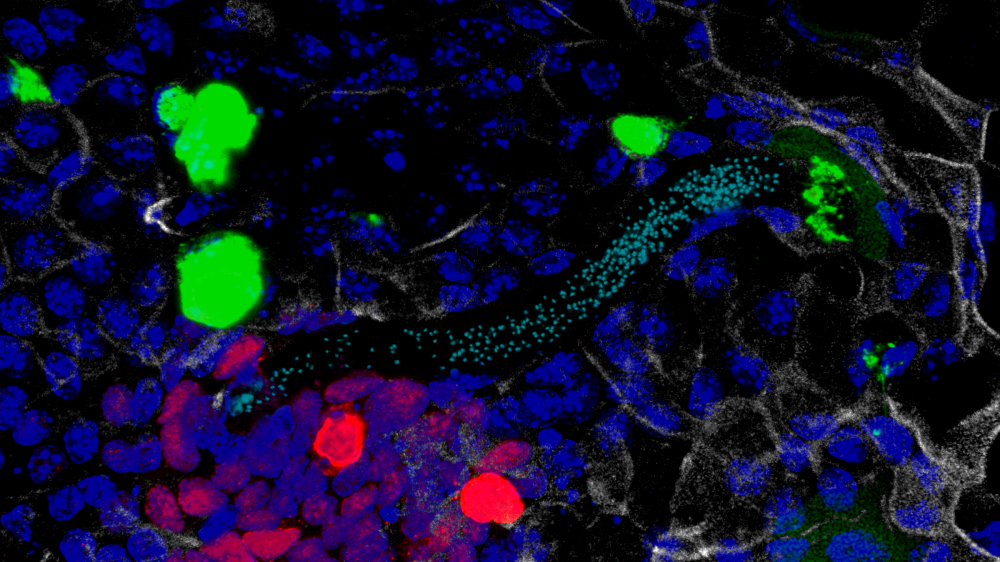
Previous studies on the immune response to whipworm have heavily relied on mouse models, but it is difficult to explore the earliest stages of infection using mice. María and colleagues developed an in vitro model using intestinal organoids, or mini-guts, which can be grown in the lab from mouse gut cells – the first time such a model has been applied to a multicellular parasite. The team were able to investigate the early stages and mechanisms of whipworm infection in more detail, finding the response of the gut lining to whipworm is different to infection with other parasites, and in fact is more similar to what happens when a virus enters the body.
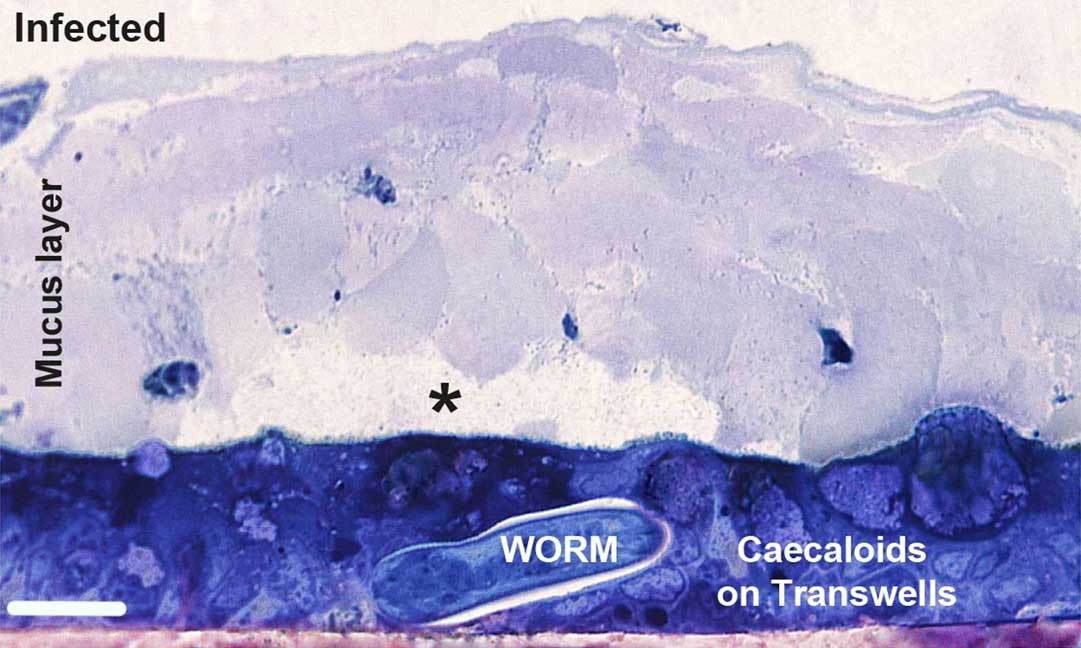
Identifying the early stages in the immune response to whipworms could help develop new treatments or vaccines against this parasite by blocking its key interactions with the host. The team previously used around 1,500 mice per year in their research on whipworms, which could be reduced to under 100 via the organoid model. The team eventually hope to apply the method to generate human cell-derived organoids that can be used to study T. trichiura in a more human-relevant model.
Dr María Duque-Correa said:
While organoids have been used to study host interactions with viruses, bacteria and protozoan parasites, this is the first time this in vitro system has been used to investigate a multicellular parasite. This model is a game changer in the trichuriasis field and will allow the reduction of the number of mice used in this research. In the future, our model can be adapted to investigate other live parasitic worms that similarly lack an in vitro experimental model, further reducing the mice used in parasitology research.
Read the full press release from the Sanger Institute: Mini guts used to study early stages of whipworm infection
Reference
- Duque-Correa MA et al. (2022). Defining the early stages of intestinal colonisation by whipworms. Nature Communications 13: e1725. doi: 10.1038/s41467-022-29334-0
Intestinal organoids as a replacement strategy to unravel early host intestinal epithelia interactions with whipworms.

Read our previous coverage of María’s public engagement work.

